In the wellness landscape of 2026, “functional water” has become a $112 billion industry. We are told to optimize our hydration with high-sodium shots and alkaline boosters, but the molecular reality is often the opposite: many of these “solutions” actually trigger an osmotic trap that pulls water out of your cells, leaving you more dehydrated than when you started.
The Evolutionary “Silent” Taste of Water
A common misconception is that humans never evolved a taste for water. In reality, our sensory physiology is tuned to the “neutrality” of pure water as a survival mechanism. By maintaining a “zero-baseline” for water, our taste buds can instantly detect life-threatening contaminants—such as sulfur, rot, or high salinity—that would be masked if water itself had a strong flavor profile.
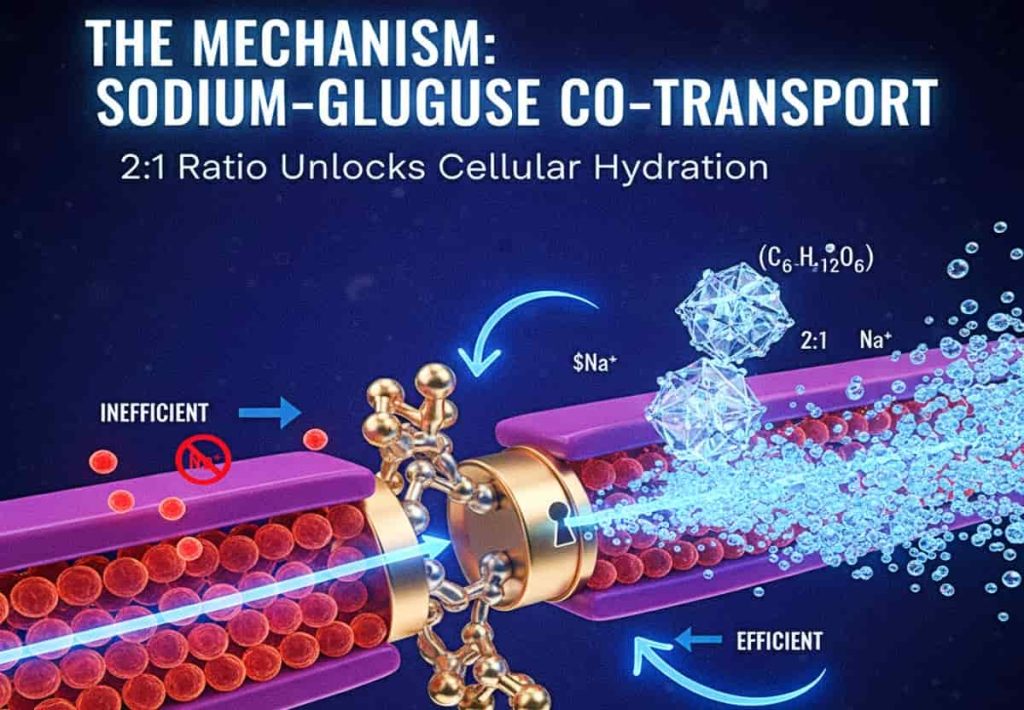
The Mechanism: The Sodium-Glucose Co-Transport
Hydration is not a passive process of “pouring water into a vessel.” It is an active transport mechanism. To move water across the intestinal wall and into the bloodstream, your body primarily uses the Sodium-Glucose Co-transport. This process requires a specific ratio of glucose to sodium (approximately 2:1) to act as the molecular “key” that opens the cellular gates for water absorption.
When you consume “wellness” waters that are overloaded with sodium but devoid of the necessary carbohydrate co-transporters, the water sits in your gut. Worse, if the concentration of salt in your intestines becomes higher than in your blood, your body will actually dump water into the gut to dilute the salt—causing “osmotic diarrhea” and acute dehydration.
Expert Map: The Hydration Evidence Hierarchy
| Hydration Source | Osmotic Efficiency | Primary Mechanism | Best Use Case |
| Plain Water | High (Maintenance) | Passive diffusion/Aquaporins | Daily sedentary hydration |
| WHO-Standard ORS | Highest (Recovery) | Sodium-Glucose Co-transport | Illness, heavy sweat, heat |
| Functional/Alkaline Water | Low to Negative | Often lacks co-transporters | Flavor only; use with caution |
| Sugary Sports Drinks | Moderate | High glucose can cause osmotic pull | Intense 60min+ athletics |
The Specialist’s Nuance: The Bioavailability Factor
Specialists point out that the recent “Alkaline Water” craze is largely built on a physiological myth. While the pH of these waters is high, they lack buffering capacity. This means that the moment they hit the hydrochloric acid in your stomach, the alkalinity is neutralized, providing zero systemic “pH balancing” benefit.
The Pro Tip: To maximize cellular hydration, look beyond the ‘alkaline’ label. Focus on your gut health, specifically the connection to Resistant Starch: How It Works, What It Helps — and What the Research Can’t Prove; a healthy gut lining with proper mucosal integrity is actually more important for electrolyte absorption than the pH of the water you drink.
FAQ
1. Can “functional water” cause high blood pressure?
Yes. Over-supplementing with high-sodium hydration powders daily can lead to sodium retention, bloating, and increased strain on the kidneys, especially in individuals who are not losing significant salt through sweat.
2. Is Molecular Hydrogen ($H_2$) the real secret to “Reduced Water”?
According to the latest clinical reviews (MDPI 2022), the therapeutic effects attributed to “ionized” or “alkaline” water are actually caused by the dissolved $H_2$ gas produced during electrolysis, which acts as a selective antioxidant.
3. Does coffee count toward my daily hydration?
While caffeine is a mild diuretic, the water in the coffee still contributes to your net hydration. However, for rapid rehydration, non-diuretic fluids like ORS or water remain superior.